Description
Overview
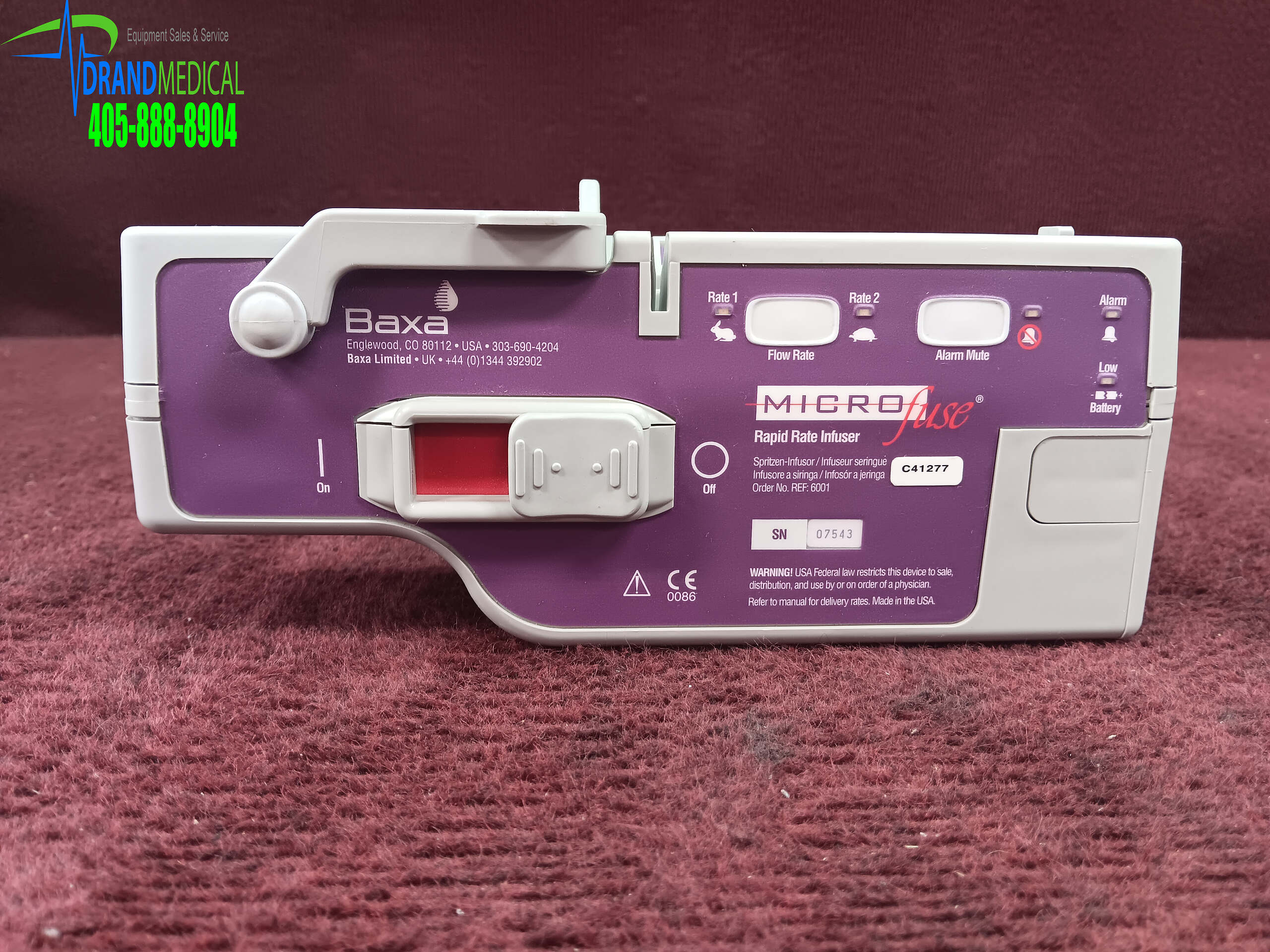
The MicroFuse® Rapid Rate Infuser is intended for
intravenous infusion of adenosine. It is designed to deliver
a “full” syringe dose over 6 minutes. The
MicroFuse Rapid Rate Infuser delivers reliable, continuous
infusion over the prescribed duration.
MicroFuse Rapid Rate Infusers are:
• Durable – solid body construction with secure motor
attachment
• Easy-to-use – simple push-button controls
• Portable – lightweight and slender profile
• Reliable – low maintenance, with built-in safety features
For Customer Support call 802-323-0101 • FAX 802-334-6971
Syringe Compatibility
The MicroFuse Rapid Rate Infuser is compatible with
Becton-Dickinson® (B-D), Monoject® and Terumo®
disposable syringes. Typical syringes for Adenoscan®
(adenosine) are B-D 30 or 60 cc, Monoject 60 cc, or
Terumo 30 or 60 cc.
The infuser operates on two standard alkaline C-cell
batteries. The syringe holder secures the syringe in place.
Once the infuser is turned ON, the syringe driver arm
depresses the plunger at a fixed rate, administering the
contents of the syringe through the extension tubing and
into the patient access site. A microprocessor control
operates at a constant delivery rate, and can infuse
syringe contents against a back pressure of ± 100 mm Hg.
The MicroFuse Rapid Rate Infuser infuses at one of two
pre-set flow rates. RATE 1 infuses a “full” syringe over 6
minutes. RATE 2 infuses a “full” syringe over 8.4 minutes.
If RATE 1 is set to deliver 140 mcg/kg/min of adenosine,
RATE 2 will slow the dose to 100 mcg/kg/min.
The MicroFuse Rapid Rate Infuser has a full complement of
alarms that signal: end of infusion, occlusion detection,
low battery, maintenance required and internal
malfunction. Alarms are indicated by flashing status lights
and an audible chime. NOTE: Audible alarms may be
placed in MUTE mode by pressing Alarm Mute switch for
two Operating Procedures
Follow accepted IV therapy techniques and procedures
when setting up and administering intermittent IV therapy
with the MicroFuse Rapid Rate Infuser. Refer to drug
manufacturer’s guidelines.
A. Setup
1. Remove air from the syringe.
2. Calculate the correct volume of adenosine for a 140
mcg/kg/min dose, using the chart provided by Astellas
Pharma Inc. or other manufacturers.
3. Draw up the adenosine solution in one of the following
ways:
a. Use a B-D 30 mL or Terumo 30 mL syringe for doses
up to 30 mL For patients up to 231 pounds at 840
mcg/kg total dose using 3 mg/mL adenosine.
b. Use a B-D or Monoject 60 mL syringe for 30 to 45 mL
doses (may be used for all doses up to 45 mL by
following step 4 below).
For patients up to 353 pounds at 840 mcg/kg
total dose using 3 mg/mL adenosine.
c. Use a Terumo 60 cc syringe for doses up to 51 mL
For patients up to 400 pounds at 840 mcg/kg total
dose using 3 mg/mL adenosine.
4. Add additional normal saline to the same syringe for:
a. 30 mL total volume in the B-D 30 mL syringe
b. 45 mL total volume in either the B-D 60 mL or
Monoject 60 mL syringe
c. 51 mL total volume in the Terumo 60 cc syringe full seconds.
B. Install Syringe
1. Load the syringe into the infuser by sliding it
underneath the syringe barrel holder. Alternately, pull
open the barrel holder and place the syringe on the infuser.
2. Position the syringe barrel flange in the syringe barrel
flange slot on the infuser body.
3. Position the syringe plunger in the slot on the syringe
driver arm.
C. Set Up Tubing
1. Attach small bore tubing to the syringe.
2. Prime the tubing